How Medical Writing Services Shapes Clinical Research, Regulatory Compliance, and Patient Communication
Medical writing are essential within the life sciences arena, converting dense clinical and scientific information into precise, user-friendly, and regulation-ready documents for agencies, clinicians, and patients alike. Whether preparing clinical study reports, regulatory submissions, peer-reviewed manuscripts, or educational materials, these professionals guarantee transparency, bolster credibility, and shorten the approval timeline.
Increasingly global clinical trial portfolios, heightened and geographically diverse regulatory expectations, and the demand for lay-friendly communication are pushing sponsors to enlarge their capabilities, prompting them to engage specialized contract research organizations offering Medical Writing Services. Looking ahead, AI-assisted content generation, seamless integration with electronic trial master files, heightened attention to pragmatic trial findings, comprehensive language support, reinforced encryption, and adherence to clinical deadlines are likely to enhance the discipline, establishing Medical Writing as a cornerstone of forward-looking clinical strategy and healthcare delivery.
What services are included in medical writing?
Medical writing is the art of drafting documents that transform intricate scientific and clinical information into straightforward and compliant documents. Medical writing spans a variety of documents such as:
Clinical Research Documents: These include protocols, informed consent documents, clinical study reports (CSRs), and case narratives, as well as investigator brochures.
Regulatory Submissions: These include Common Technical Document (CTD) modules, Investigational New Drug (IND) applications, New Drug Applications (NDAs), Biologics License Applications (BLAs), and post-marketing safety reports.
Scientific Publications: These include reports, conference talks, as well as abstracts and posters, along with peer-reviewed manuscripts.
Healthcare Communications: These includes patient education and advocacy resources, as well as medical marketing and continuing medical education (CME) materials.
Importance of Medical Writing in Clinical Research
Medical writing does not merely involve composing documents; it’s a complex discipline that entails the triangulation of certain elements to ensure the final product is:
Accurate: Preserving scientific truth while upholding the integrity of the discipline.
Compliant: Instructions from ICH, FDA, EMA, GCP, and other international authorities must be met.
Clear: Translating and simplifying scientific jargon so that patients and other non-specialists can grasp the message.
Credible: Justifying a regulatory decision on the approval of a drug, a clinical practice guideline, or a health policy.
Medical writing serves to connect the data produced in clinical trials with the information submitted to regulators, doctors, and the wider public. Without accurate and complete documents, a new drug or therapy, or a medical device is likely to be unduly delayed, misinterpreted, or poorly complied with in the approval process.
Growing Global Demand for Medical Writing
In the pharmaceutical, biotechnology, Contract research Organizations, and wider medical sectors, there is a growing need for medical documents that meet the rigorous standards of accuracy and quality. This need is fueled by the following factors:
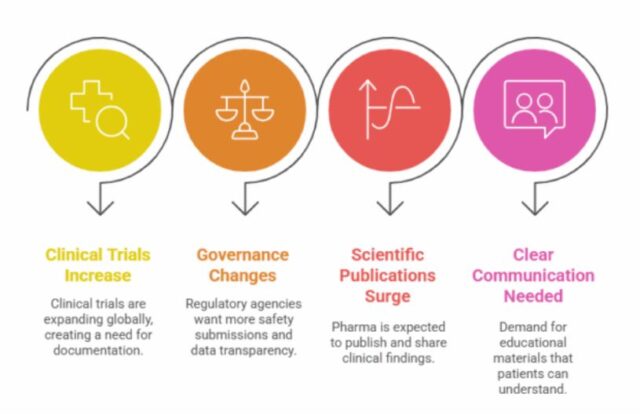
- Increasing the Number of Clinical Trials: As trials are expanding globally, there is a need for uniform and regulatory-compliant documentation.
- Changing Landscape of Governance: After marketing a product, regulatory agencies are demanding more comprehensive submissions on safety, along with clinical data transparency.
- The Surge in Scientific Publications: Regulatory bodies and stakeholders expect the pharma and biotech industry to publish actively and disseminate clinical findings.
- The Need for Clear Communication in Medicine: There is an increasing demand for educational and promotional materials that are easy for patients and the general public to understand.
Outsourcing has become a distinguishing trend for several pharma and biotech companies which contract the medical writing to cost-effective and efficient specialists like Contract research Organizations.
In the research and development department, the further advanced a country is, the more there is a flourishing complex of interdependent health professionals on a regional and international level. They seek value for their money, and their focus is largely data-driven. The market continues to require and value professional medical writing, which is a further testament to its growing significance.
Choosing the right medical writing services partner involves assessing several crucial elements:
Writer Credentials – Prioritize teams whose writers hold advanced life-science degrees, accumulative years in clinical research, and a well-honed grasp of protocol design and submission norms.
Professional Affiliations – Active membership in organizations such as AMWA, EMWA, or ISMPP, along with applicable credentials, signals an adherence to evolving industry standards.
Experience Depth – Assess a comprehensive portfolio that includes clinical study reports, regulatory dossiers, and peer-reviewed manuscripts, ensuring versatility across document formats.
Compliance History – Verify that the provider has maintained an unblemished record with regulatory bodies such as the FDA, EMA, and ICH, affirming their ability to uphold global submission expectations.
Client References – Collect and review testimonials and structured success narratives to gauge consistent service quality, punctuality, and responsiveness.
Tailored Solutions – Confirm the ability to create bespoke document development plans for varied deliverables, such as protocols, clinical study reports, or regulatory summaries, across the desired therapeutic area.
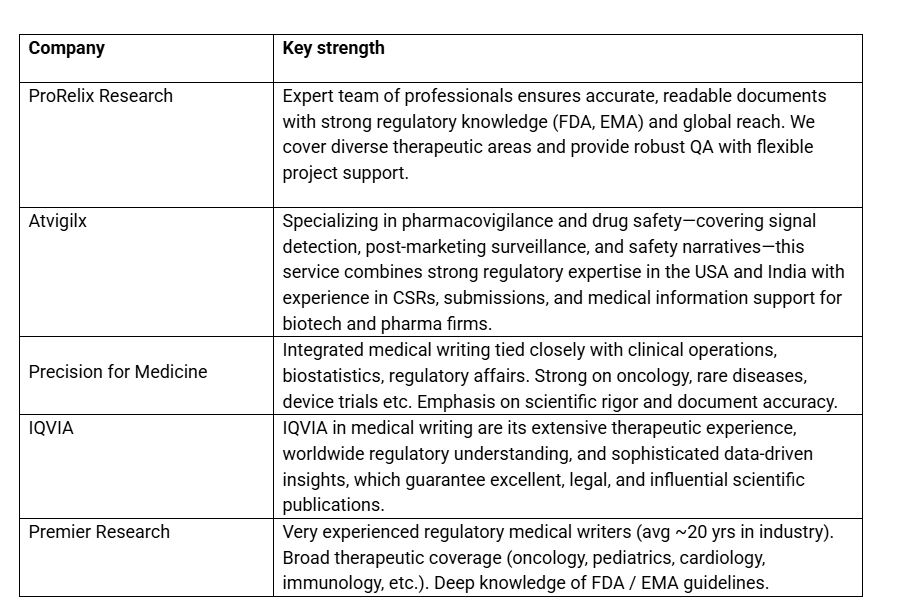
List of Top Medical writing services in USA
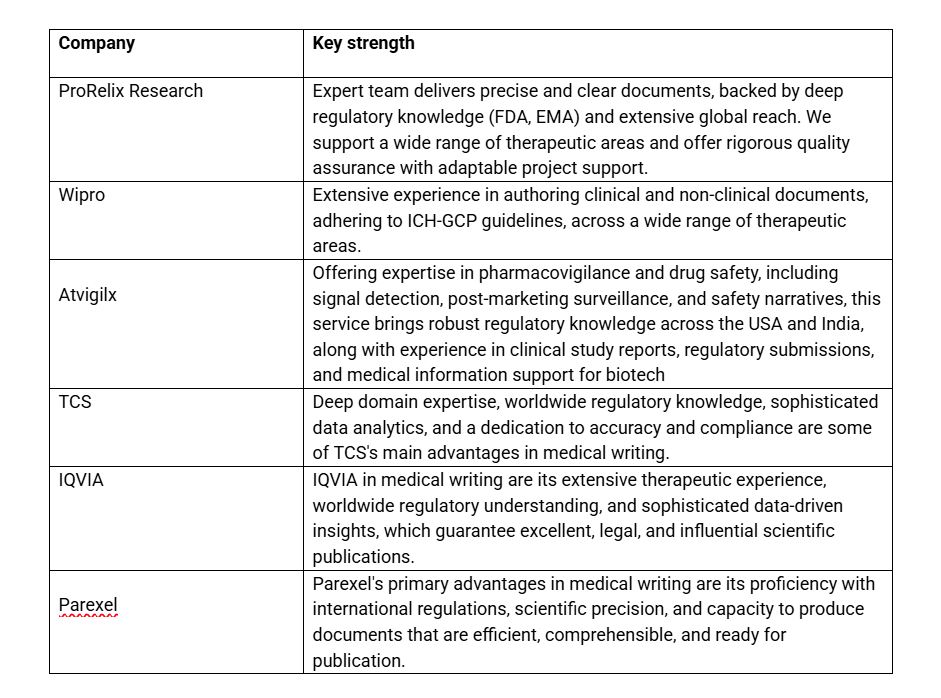
List of Top Medical writing services in India
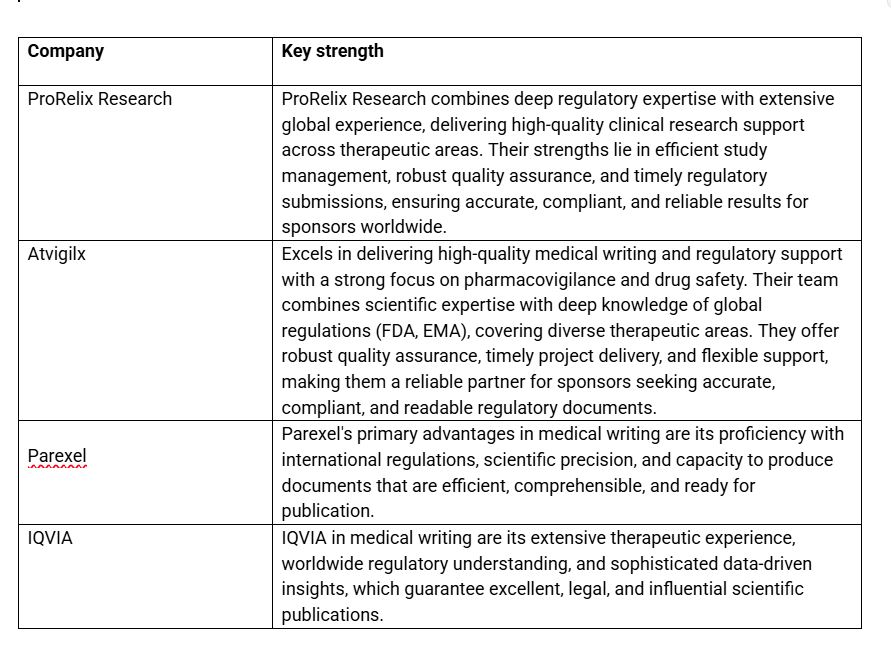
List of Top Medical writing services in Europe
Future Trends in Medical Writing
Today’s medical writing sector is continually adapting to support complex clinical programs, regulatory needs, and healthcare communications spanning the globe. Five interlinking trends are defining the next decade:
AI Utilization: Generative and analytical AI is being embedded to streamline time-consuming tasks, synthesise literature, and help draft regulatory and scientific documents. The technology is elevating productivity while maintaining stricter compliance and accuracy.
Decentralized Integration: Medical writing is converging with eSource and eRegulatory platforms to perform near real-time updates. By bringing eSource, regulatory, and writing functions together on single digital systems, the sector is minimising transcription error and latency.
Evolving Evidence Requirements: Regulatory and payer bodies are further prioritising real-world evidence. As clinical effectiveness and safety need translation into practical healthcare insights, medical writers are being tasked with collating, formatting, and contextualising data in increasingly complex RWE deliverables.
Worldwide Multilingual Coverage: Thought leaders, regulators, and trial participants now span wider geographies, compelling writing teams to create documents aligned with multilingual, regulatory, and culturally specific norms while maintaining fidelity.
Data Protection and Regulatory Rigor: Compounding the above, sponsors demand that documents, systems, and personnel are designed to guard patient identity and trial integrity. Writing teams are consequently strengthening encryption, audit trails, and procedural controls in already-stressed workflows.
Accelerated Turnaround: The combined pressures of data regulators, sponsors, and trial stakeholders are shrinking acceptable timelines. Teams are countering by adopting advanced project management software, extensive template libraries, and synchronous co-authoring tools while maximising inter-discipline collaboration.
Medical writing has evolved beyond the realm of routine documentation—it is now a vital strategist accelerating clinical development while enhancing safety and transparency across the entire process.
Conclusion
Today’s life sciences architecture rests firmly upon the platform of professional medical writing services, which seamlessly convert intricate clinical data, regimented regulatory demands, and targeted healthcare messaging into coherent narratives. This expertise manifests from the meticulous crafting of clinical trial protocols and regulatory submissions, to the rigorous drafting of scientific manuscripts and the clear presentation of patient educational materials—every document serving to optimize accuracy, uphold compliance, and illuminate understanding throughout the drug and device lifecycle.
Renewed momentum in global clinical research, compounded by spirited initiatives in data transparency, real-world evidence, and patient-centric discourse, propels ever-increasing demand, particularly from leading hubs across the USA, India, and Europe. Organizations that engage seasoned, credentialed, and visionary medical writing professionals consequently secure a competitive edge, expedited approvals, and enhanced confidence among regulators, healthcare professionals, and patients.
As capabilities expand to include adaptive AI tools, multilingual delivery, and analytics-anchored methodologies, the discipline evolves from optional adjunct to fundamental enabler of compliance, innovation, and assured market navigation. Selecting an aligned medical writing partner is therefore among the most consequential strategic choices available, capable of steering entire clinical programs toward heightened certainty, expedited timelines, and demonstrably improved patient outcomes across the globe.
FAQ’S
1) What are medical writing services?
Answer: Medical writing services produce concise, precise, and scientifically validated documents tailored for the healthcare and life sciences sectors. Offerings span regulatory submissions, clinical study documents, safety reports, peer-reviewed publications, and educational resources, promoting accurate dissemination of intricate medical information in accordance with worldwide regulatory and ethical standards.
2) How much does medical writing cost?
Answer : Medical writing fees differ considerably based on expertise, document type, and geographic area. In the United States, skilled medical writers generally invoice between $125 and $155 for each hour worked, or between $0.57 and $1.00 per word. Freelance marketplaces such as Upwork feature ranges of $23 to $50 per hour for less-experienced personnel. In India, lower overhead translates to fees of ₹2 to ₹5 per word for standard work, rising to ₹8 to ₹20 per word when projects demand specialized proficiency. Additional determinants include the document complexity, deadlines, and the level of subject matter familiarity required of the writer.
3) How stressful is medical writing?
Answer : Medical writing can be stressful, particularly within regulatory and safety functions where sharp deadlines and intricate content rule the day. Over time, cadence-born judgment, well-coordinated collaboration, and disciplined foresight level the heat. In contrast, functions anchored in broader medical communications typically enjoy a milder ride.
4) What are the different types of medical writing?
Answer : Medical writing involves creating clear, accurate scientific content for various audiences. It includes regulatory documents (protocols, study reports), clinical and scientific writing (CRFs, journal articles), medical communications (CME materials, reviews), promotional content (brochures, websites), patient-focused materials (leaflets, guides), and technical/device manuals, each tailored to its audience.
Recent Posts
- Top 10 Literature Screening and Review Service Providers in USA June 5, 2026
- Risk Management Plans Integration in Global Development Programs March 31, 2026
- Post-Marketing Surveillance (PMS) in Regulatory Affairs: A Complete Guide March 24, 2026
- PBRER vs PSUR: Key Differences Explained March 16, 2026
- Global Signal Detection Trends Transforming Drug Safety 2026 March 4, 2026

