
Abstract
As the pharmaceutical landscape becomes increasingly complex in 2026, global signal detection is emerging as a critical component of drug safety. Advanced therapies, biologics, and personalized medicine require proactive monitoring strategies that go beyond traditional approaches. This review examines the key trends shaping pharmacovigilance, including artificial intelligence, real-world evidence, predictive analytics, and global collaboration. By highlighting opportunities, challenges, and future directions, it provides a comprehensive view of how signal detection is transforming drug safety worldwide.

Why Global Signal Detection is Essential?
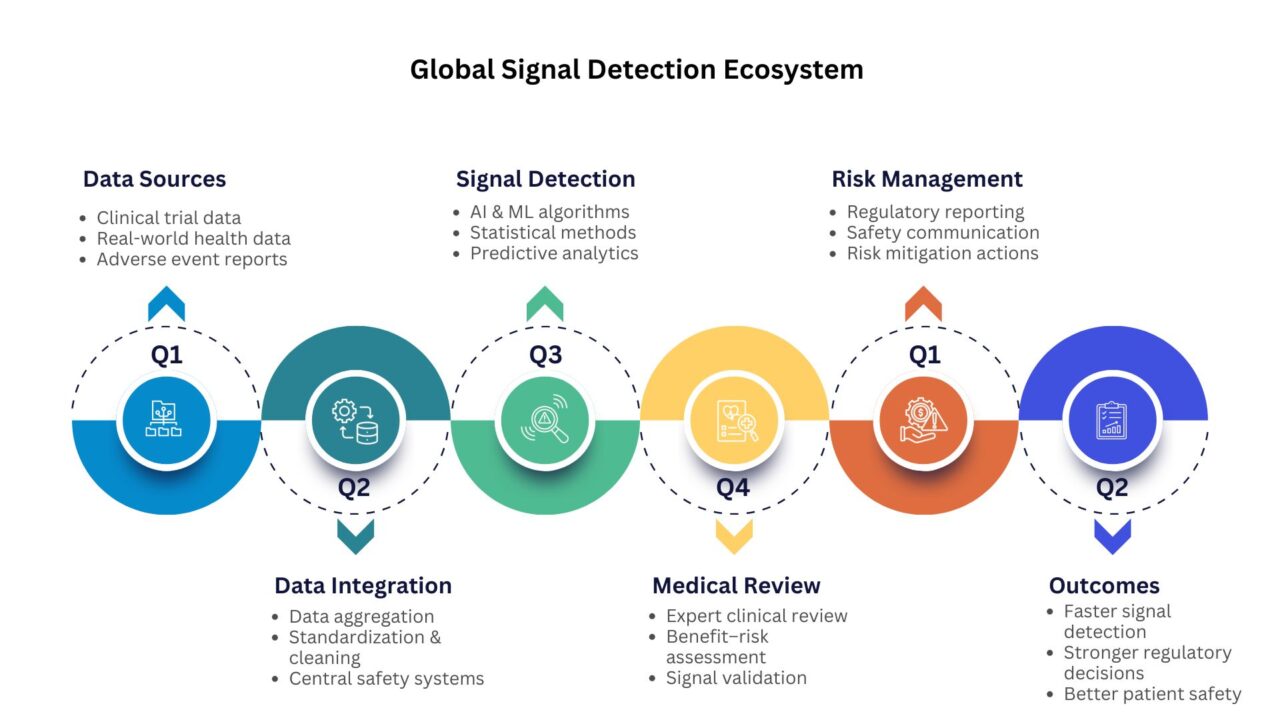
Global Signal Detection is essential because it enables early identification of potential drug safety risks, supports regulatory decision-making, enhances patient protection, and strengthens post-marketing surveillance through continuous analysis of global safety data.
Building on this foundation, signal detection is the systematic process of identifying potential safety concerns from multiple data sources. In modern pharmacovigilance, it serves several critical purposes:
Detecting emerging safety risks early
Supporting regulatory decision-making with comprehensive evidence
Enhancing patient safety through timely interventions
Optimizing risk management and post-marketing surveillance through continuous monitoring of real-world safety data.
The shift toward global signal detection allows for integration of safety data across multiple geographies, treatment settings, and patient populations. This approach provides a more complete picture of a drug’s safety profile, ensuring risks are identified promptly and acted upon effectively.
Key Trends Transforming Signal Detection
1. Artificial Intelligence and Machine Learning
Artificial intelligence and machine learning are transforming the speed and accuracy of signal detection. Advanced algorithms can process millions of data points from electronic health records, clinical trial data, literature, patient registries, and social media.
Machine learning models continuously improve over time, identifying subtle patterns that may escape traditional analysis. This proactive capability is particularly valuable for complex therapies, including gene and cell-based treatments, where adverse events may be rare or multifactorial.
2. Real-World Evidence Integration
Real-world evidence (RWE) has become a vital component of pharmacovigilance. By integrating data from electronic health records, insurance claims, and patient-reported outcomes, organizations can evaluate drug safety in routine clinical practice across diverse populations.
RWE complements clinical trial data, providing insights into long-term outcomes, off-label use, and population-specific risks. Regulatory agencies are increasingly recognizing its value, using Real- world evidence(RWE) to support label updates, post-marketing safety evaluations, and risk management decisions.
3. Standardization and Global Data Harmonization
Global signal detection requires consistent, high-quality data. Differences in reporting standards, terminologies, and coding practices across regions can hinder analysis. Standardization, using tools such as MedDRA coding and guidelines from the International Council for Harmonisation (ICH), ensures data from multiple sources can be aggregated and interpreted accurately.
Table: Benefits of Global Data Harmonization
| Challenge Without Harmonization | How Harmonization Addresses It | Outcome |
| Inconsistent terminology across regions | Standard coding systems such as MedDRA | Accurate signal detection |
| Data fragmentation from multiple sources | Structured integration frameworks | Comprehensive risk assessment |
| Regulatory review delays | Global alignment of reporting standards | Faster regulatory decision-making |
| Redundant or missed signals | Unified datasets and analytics | Improved patient safety |
4. Predictive Analytics and Proactive Risk Management
Predictive analytics allows organizations to anticipate potential safety risks. By analyzing historical data, patient demographics, and drug usage patterns, teams can prioritize high-risk signals and allocate resources efficiently.
This proactive approach reduces the reliance on reactive monitoring and supports evidence-based risk mitigation strategies, ultimately safeguarding patient health while optimizing operational efficiency.
5. Collaborative Global Surveillance Networks
The increasing complexity of global clinical trials and post-marketing surveillance has driven collaboration among pharmaceutical companies, regulators, and academic institutions. These international networks enable sharing of safety data, best practices, and analytical tools, accelerating signal detection and improving reliability.
Collaboration ensures emerging risks are addressed promptly, enhances regulatory compliance, and strengthens public confidence in therapeutics.
Challenges in 2026
Despite technological advancements, several challenges remain:
- Data Privacy and Compliance: Regulations such as GDPR require careful handling of patient data, particularly when integrating multi-country datasets.
- Data Integration Complexity: Combining structured and unstructured data from diverse sources introduces noise and variability.
- Validation of AI Models: Continuous validation is required to prevent false positives or negatives that could impact patient safety.
- Workforce Expertise: Skilled professionals are essential to interpret complex analytics, implement strategies, and maintain global collaboration.
Addressing these challenges is essential to fully realize the potential of global signal detection and ensure its effectiveness in protecting patient health.
Future Outlook
Looking ahead, global signal detection will become more predictive, proactive, and patient-centric. Key developments expected in 2026 include:
Greater adoption of AI-driven real-time monitoring systems for rapid signal identification
Expansion of real-world evidence integration to support regulatory decision-making and post-marketing safety
Stronger international collaboration to ensure timely detection of emerging risks across regions
Clearer regulatory guidance on the use of AI and advanced analytics in pharmacovigilance
These trends suggest that drug safety monitoring will increasingly move from reactive compliance to strategic, data-driven risk management, benefiting both patients and healthcare systems.
Conclusion
Global signal detection is fundamentally transforming drug safety in 2026. By combining advanced analytics, real-world evidence, standardized data practices, predictive insights, and global collaboration, pharmacovigilance is becoming more proactive, accurate, and patient-focused.
Organizations that embrace these trends can ensure timely identification and mitigation of risks, streamline regulatory compliance, and improve patient outcomes. The evolution of signal detection represents not just a technological shift but a strategic opportunity to redefine the future of drug safety worldwide.
Read More: How Signal Pharmacovigilance Protects Patient Safety: Navigating Risks
Frequently Asked Questions (FAQs)
What is global signal detection in pharmacovigilance?
How does artificial intelligence improve signal detection?
What role does real-world evidence play in drug safety monitoring?
Why is data harmonization important in global signal detection?
What are the key challenges in modern signal detection?
Recent Posts
- Global Signal Detection Trends Transforming Drug Safety 2026 March 4, 2026
- How Medical Writing Services Shapes Clinical Research, Regulatory Compliance, and Patient Communication November 6, 2025
- EudraVigilance Setup & Maintenance – Smart, Scalable Solutions June 6, 2025
- Benefit Risk Management in Pharmacovigilance for Better Drug Safety April 17, 2025
- Safety Management Plan in Pharmacovigilance for Pharma Companies Ensuring Safety April 15, 2025

